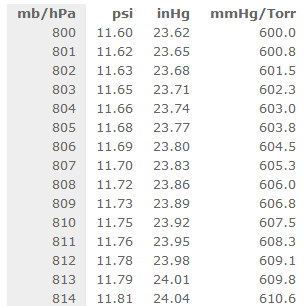
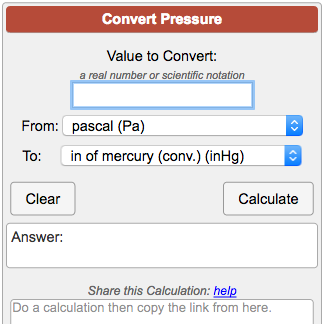
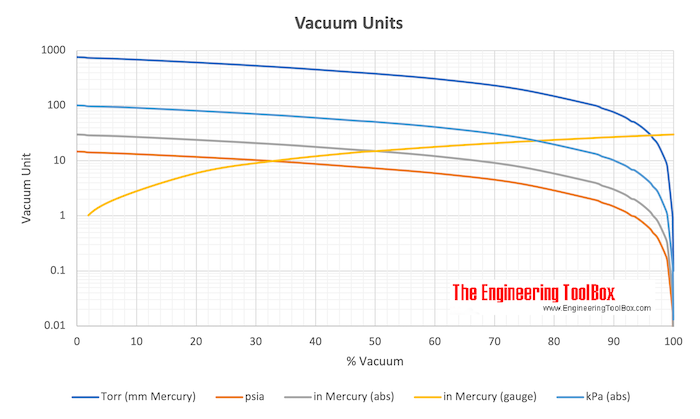
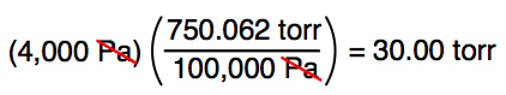Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download
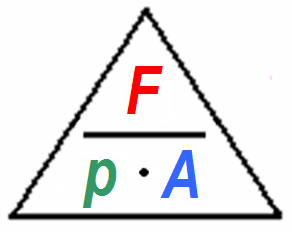
Conversion of pressure units psi bars atm pressure Pa force F Newton divided by area A psi pascals bars atm - sengpielaudio Sengpiel Berlin

SOLVED:The pressure on top of Mount Everest (29,029 ft or 8848 m) averages about 235 mmHg. Convert this pressure to \begin{equation}\begin{array}{ll}{\text { a. torr }} & {\text { b. psi }} \\ {\